Frequently Asked Questions
Li-ion batteries are rechargeable batteries that use lithium ions which move from a negative electrode to a positive electrode. They are popular batteries for use in consumer electronics because they provide good energy density, no memory effect, and a slow loss of charge when not in use. These batteries come in a wide variety of shapes and sizes. In addition, compared to lead-acid batteries, Li-ion batteries are lighter and provide a higher open circuit voltage, which allows for power transfer at lower currents. These batteries have the following characteristics:
- Enhanced safety
- Exceptional low temperature performance
- Good cycle life
- High abuse tolerance*
- High capacity
- High rate capability
- High temperature performance
*Applies to LiFEPO4 only, does not apply to Cobalt Oxide or Lithium Polymer
The term "Li-ion" battery is a general term like the term "car" and refers to many different types of Lithium. There are many different chemistries for Li-Ion batteries including LiCoO2 (cylindrical cell), LiPo, and LiFePO4 (cylindrical/prismatic cell). ToBattery.com currently is focusing on design, manufacturing, and marketing of LiFePO4 (LFP) batteries.
The different types of Li-ion batteries have different characteristics. Typically when the term "Li-ion" is used in a product description what is being sold may be either LiCoO2 (Cobalt Oxide) or LiPo (Lithium Polymer) unless LiFePO4 (Lithium Iron Phosphate) is specifically named. This is the rule of thumb because LiCoO2 and LiPo are the most common types of Lithium. The LiCoO2 in particular is cheaper and has a high power density but as a trade off has drastically shortened service life and is chemically and thermally unstable. Within this lineup LiFePO4 is unique for its aforementioned performance benefits. Compared with the older LiCoO2, LiPo and other chemistry such as AGM, SLA and NiCad, the current state-of-the-art LiFePO4 is essentially a new and entirely different chemistry unique unto itself.
LiPo batteries are a type of Li-Ion battery used for a variety of high current discharge applications such as in RC toy models (cars, helicopters, airplanes, boats, etc.). They are also used in various consumer electronics because of their compact size.The LiPo batteries are comprised of cells that are stacked in series/parallel configurations. Each cell contains anode and cathode plates with a gel electrolyte that are placed in a pouch. These batteries are low in costs, can be packaged into a variety of shapes (which is important for use in electronics such as cell phones), light in weight, and rugged.
Lithium Iron Phosphate (LiFePO4) batteries use a new type of cathode material that provides several advantages over traditional Li-ion batteries based on LiCoO2. LiFePO4 batteries provide much higher specific capacity, superior thermal and chemical stability, enhanced safety, improved cost performance, enhanced charge and discharge rates, compact size, light weight, and enhanced cycle life. LiFePO4 batteries can offer a cycle life of potentially 2,000-3,000 charge cycles. Typical LFP batteries have an average lifetime of between 5 to 10 years and can be expected to deliver reliable and consistent performance throughout its service life. The characteristics Bioenno Power batteries are proven to offer are as such:
- Enhanced safety
- Exceptional low temperature performance
- Enhanced 2000-3000+ charge cycles
- Enhanced 5-10+ years service life
- High abuse tolerance
- High capacity - a Bioenno Power LFP will generally outperform an SLA with twice its Ah rating.
- High rate capability
- High temperature performance
- Chemical and Thermal stability
- Flat, consistent discharge
- No Memory Effect
- Full depth discharge
- No need for tendering of floating
LiFePO4 batteries themselves come in two distinct types as determined by cell type:
- Deep Cycle LiFePO4: essentially the distinction boils down to the amount of lithium present in each cell and the intended application which in this case is to power electronics for long periods of time. This type is designed with capacity in mind and at its heart stores lots of power in a very small package and as such has more lithium per cell. A deep cycle battery of this chemistry will not be able to crank your engine.
- High Rate LiFePO4: these are for starting motors only, the difference being that this type has dramatically lower lithium content per cell. However, the lower lithium quantity is not an issue because by design this type is only intended to output large bursts of energy for a short amount of time. As such, this type does not need that much lithium. While this type can be mistakenly used for a deep cycle application, doing so incurs significant risk as the high current rating has a high likelihood of damaging your load. These batteries are easy to identify because they have two distinct characteristics: A) a Lead Acid Equivalent Figure and B) have a CCA (Cold Cranking Amp) Rating.
These two types are not to be confused or interchanged. While a High Rate will be able to power electronics, it will only do so momentarily as they by design do not have the storage capacity needed for extended use. Moreover, the high current ceiling will not mitigate current spikes the occurrence of which will damage your equipment. If you are powering equipment, you require Deep Cycle Batteries. If you are starting motors or powering your flash on your camera, the High Rate is the one for you. Remember the following rule of thumb:
Deep cycle/deep discharge and high rate/starter are two different batteries even if they are from the same chemistry. This holds true for all battery types, even Lead Acid/SLA/GEL/AGM. A battery is either for deep cycle or for starting, never both despite any claims which may be made so make sure to double check your application and confirm your requirements before purchase.
In essence these are a type of single-board microcomputer. PCMs, or protection circuit modules, are a type of battery management system (BMS) for the expressed purpose of regulating output of current from the battery to electrical loads, managing and internally balancing the cell pack in addition to regulating charging. They are there to protect the cells from damage. If your electric load exceeds the power load of the battery the PCM shuts the battery off to protect the cells from damage. This is a safeguard to preserve maximum functionality of our batteries and to, effectively, abuse-proof within a certain extent. Our entire range of batteries all come equipped with top of the line sophisticated PCMs unlike a large number of our competitors. We tested our products. It is almost impossible to screw up our batteries because our integrated intelligent systems automatically shuts off the battery before damage can be done to it. Our PCMS offer the following features:
- Balancing function for cells
- Overcurrent protection
- Overvoltage protection
- Overdischarge protection (also known as undervoltage)
- Temperature protection
- Short circuit protection
- Charging Circuitry integrated
Compare the pros and cons of the cylindrical cell, button cell, prismatic cell and pouch
As batteries were beginning to be mass-produced, the jar design changed to the cylindrical format. The large F cell for lanterns was introduced in 1896 and the D cell followed in 1898. With the need for smaller cells, the C cell followed in 1900, and the popular AA was introduced in 1907. See BU-301: Standardizing Batteries into Norms.
Cylindrical Cell
The cylindrical cell continues to be one of the most widely used packaging styles for primary and secondary batteries. The advantages are ease of manufacture and good mechanical stability. The tubular cylinder can withstand high internal pressures without deforming.
Many lithium and nickel-based cylindrical cells include a positive thermal coefficient (PTC) switch. When exposed to excessive current, the normally conductive polymer heats up and becomes resistive, stopping current flow and acting as short circuit protection. Once the short is removed, the PTC cools down and returns to the conductive state.
Most cylindrical cells also feature a pressure relief mechanism, and the simplest design utilizes a membrane seal that ruptures under high pressure. Leakage and dry-out may occur after the membrane breaks. Re-sealable vents with a spring-loaded valve are the preferred design. Some consumer Li-ion cells include the Charge Interrupt Device (CID) that physically and irreversibly disconnect the cell when activated to an unsafe pressure builds up. Figure 1 shows a cross section of a cylindrical cell.
|
Figure 1: Cross section of a lithium-ion cylindrical cell. |
Typical applications for the cylindrical cell are power tools, medical instruments, laptops and e-bikes. To allow variations within a given size, manufacturers use partial cell lengths, such as half and three-quarter formats, and nickel-cadmium provides the largest variety of cell choices. Some spilled over to nickel-metal-hydride, but not to lithium-ion as this chemistry established its own formats. The 18650 illustrated in Figure 2 remains one of the most popular cell packages. Typical applications for the 18650 Li-ion are power tools, medical devices, laptops and e-bikes.
|
Figure 2: Popular 18650 lithium-ion cell. |
In 2013, 2.55 billion 18650 cells were produced. Early Energy Cells had 2.2Ah; this was replaced with the 2.8Ah cell. The new cells are now 3.1Ah with an increase to 3.4Ah by 2017. Cell manufacturers are preparing for the 3.9Ah 18650.
The 18650 could well be the most optimized cell; it offers one of the lowest costs per Wh and has good reliability records. As consumers move to the flat designs in smart phones and tablets, the demand for the 18650 is fading and Figure 3 shows the over-supply that is being corrected thanks to the demand of the Tesla electric vehicles that also uses this cell format for now. As of end of 2016, the battery industry fears battery shortages to meet the growing demand for electric vehicles.
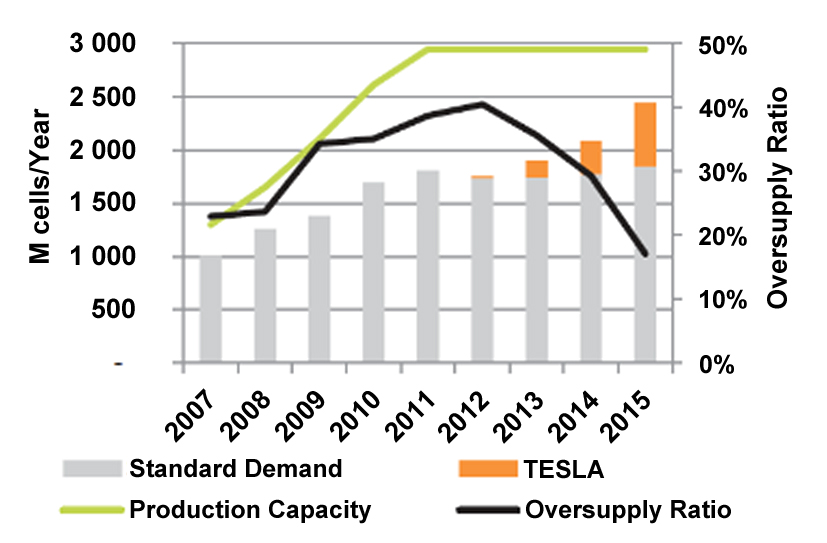 |
| Figure 3: Demand and supply of the 18650. The demand for the 18650 would have peaked in 2011 had it not been for new demands in military, medical and drones, including the Tesla electric car. The switch to a flat-design in consumer products and larger format for the electric powertrain will eventually saturate the 18650. A new entry is the 21700. Source: Avicenne Energy |
There are other cylindrical Li-ion formats with dimensions of 20700, 21700 and 22700. Meanwhile, Tesla, Panasonic and Samsung have decided on the 21700 for easy of manufacturing, optimal capacity and other benefits. While the 18650 has a volume of approximately 16cm3 (16ml) with a capacity of around 3000mAh, the 21700 cell has approximately 24cm3 (24ml) with a said capacity of up to 6000mAh, essentially doubling the capacity with a 50% increase in volume. Tesla Motor refers to their company’s new 21700 as the “highest energy density cell that is also the cheapest.” (The 2170 nomenclature Tesla advocates is not totally correct; the last zero of the 21700 model describes a cylindrical cell harmonizing with the IEC standard.)
The larger 26650 cell with a diameter of 26mm does not enjoy the same popularity as the 18650. The 26650 is commonly used in load-leveling systems. A thicker cell is said to be harder to build than a thinner one. Making the cell longer is preferred. There is also a 26700 made by E-One Moli Energy.
Some lead acid systems also borrow the cylindrical design. Known as the Hawker Cyclone, this cell offers improved cell stability, higher discharge currents and better temperature stability compared to the conventional prismatic design. The Hawker Cyclone has its own format.
Even though the cylindrical cell does not fully utilize the space by creating air cavities on side-by-side placement, the 18650 has a higher energy density than a prismatic/pouch Li-ion cell. The 3Ah 18650 delivers 248Ah/kg, whereas a modern pouch cell has about 140Ah/kg. The higher energy density of the cylindrical cell compensates for its less ideal stacking abilities and the empty space can always be used for cooling to improve thermal management.
Cell disintegration cannot always be prevented but propagation can. Cylindrical cells are often spaced apart to stop propagation should one cell take off. Spacing also helps in the thermal management. In addition, a cylindrical design does not change size. In comparison, a 5mm prismatic cell can expand to 8mm with use and allowances must be made.
Button Cell
The button cell, also known as coin cell, enabled compact design in portable devices of the 1980s. Higher voltages were achieved by stacking the cells into a tube. Cordless telephones, medical devices and security wands at airports used these batteries.
Although small and inexpensive to build, the stacked button cell fell out of favor and gave way to more conventional battery formats. A drawback of the button cell is swelling if charged too rapidly. Button cells have no safety vent and can only be charged at a 10- to 16-hour charge; however, newer designs claim rapid charge capability.
Most button cells in use today are non-rechargeable and are found in medical implants, watches, hearing aids, car keys and memory backup. Figure 4 illustrates the button cells with a cross section.
| CAUTION | Keep button cells to out of reach of children. Swallowing a cell can cause serious health problems. See BU-703 Health Concerns with Batteries. |
|
|
Figure 4: Button cells provides small size, most are primary for single-cell use.
Source: Sanyo and Panasonic
Prismatic Cell
Introduced in the early 1990s, the modern prismatic cell satisfies the demand for thinner sizes. Wrapped in elegant packages resembling a box of chewing gum or a small chocolate bar, prismatic cells make optimal use of space by using the layered approach. Other designs are wound and flattened into a pseudo-prismatic jelly roll. These cells are predominantly found in mobile phones, tablets and low-profile laptops ranging from 800mAh to 4,000mAh. No universal format exists and each manufacturer designs its own.
Prismatic cells are also available in large formats. Packaged in welded aluminum housings, the cells deliver capacities of 20–50Ah and are primarily used for electric powertrains in hybrid and electric vehicles. Figure 5 shows the prismatic cell.
|
Figure 5: Cross section of a prismatic cell. |
The prismatic cell requires a firm enclosure to achieve compression. Some swelling due to gas buildup is normal, and growth allowance must be made; a 5mm (0.2”) cell can grow to 8mm (0.3”) after 500 cycles. Discontinue using the battery if the distortion presses against the battery compartment. Bulging batteries can damage equipment and compromise safety.
Pouch Cell
In 1995, the pouch cell surprised the battery world with a radical new design. Rather than using a metallic cylinder and glass-to-metal electrical feed-through, conductive foil-tabs were welded to the electrodes and brought to the outside in a fully sealed way. Figure 6 illustrates a pouch cell.
|
| Figure 6: The pouch cell. The pouch cell offers a simple, flexible and lightweight solution to battery design. Some stack pressure is recommended but allowance for swelling must be made. The pouch cells can deliver high load currents but it performs best under light loading conditions and with moderate charging. Source: A123 |
The pouch cell makes most efficient use of space and achieves 90–95 percent packaging efficiency, the highest among battery packs. Eliminating the metal enclosure reduces weight, but the cell needs support and allowance to expand in the battery compartment. The pouch packs are used in consumer, military and automotive applications. No standardized pouch cells exist; each manufacturer designs its own.
Pouch packs are commonly Li-polymer. Small cells are popular for portable applications requiring high load currents, such as drones and hobby gadgets. The larger cells in the 40Ah range serve in energy storage systems (ESS) because fewer cells simplify the battery design.
Although easily stackable, provision must be made for swelling. While smaller pouch packs can grow 8–10 percent over 500 cycles, large cells may expand to that size in 5,000 cycles. It is best not to stack pouch cells on top of each other but to lay them flat, side by side or allow extra space in between them. Avoid sharp edges that can stress the pouch cells as they expand.
Extreme swelling is a concern. Users of pouch packs have reported up to 3 percent swelling incidents on a poor batch run. The pressure created can crack the battery cover, and in some cases, break the display and electronic circuit boards. Discontinue using an inflated battery and do not puncture the bloating cell in close proximity to heat or fire. The escaping gases can ignite. Figure 7 shows a swollen pouch cell.
|
Figure 7: Swollen pouch cell. |
Pouch cells are manufactured by adding a temporary “gasbag” on the side. Gases escape into the gasbag while forming the solid electrolyte interface (SEI) during the first charge. The gasbag is cut off and the pack is resealed as part of the finishing process. Forming a solid SEI is key to good formatting practices. Subsequent charges should produce minimal gases, however, gas generation, also known as gassing, cannot be fully avoided. It is caused by electrolyte decomposition as part of usage and aging. Stresses, such as overcharging and overheating promote gassing. Ballooning with normal use often hints to a flawed batch.
The technology has matured and prismatic and pouch cells have the potential for greater capacity than the cylindrical format. Large flat packs serve electric powertrains and Energy Storage System (ESS) with good results. The cost per kWh in the prismatic/pouch cell is still higher than with the 18650 cell but this is changing. Figure 8 compares the price of the cylindrical, prismatic and pouch cells, also known as laminated. Flat-cell designs are getting price competitive and battery experts predict a shift towards these cell formats, especially if the same performance criteria of the cylindrical cell can be met.
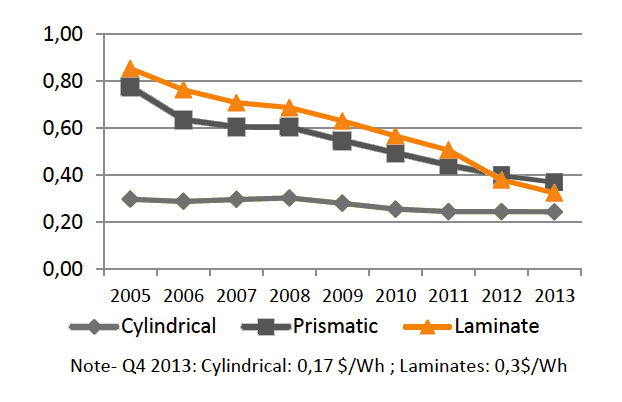 |
| Figure 8: Price of Li-ion ($US/Wh). Historically, manufacturing costs of prismatic and pouch formats (laminate) were higher, but they are converging with cellular design. Pricing involves the manufacturing of the bare cells only. Source: Avicenne Energy |
Asian cell manufacturers anticipate cost reductions of the four most common Li-ion cells, which are the 18650, 21700, prismatic and pouch cells. The 21700 promises the largest cost decrease over the years and economical production, reaching price equilibrium with the pouch by 2025 (Figure 9).
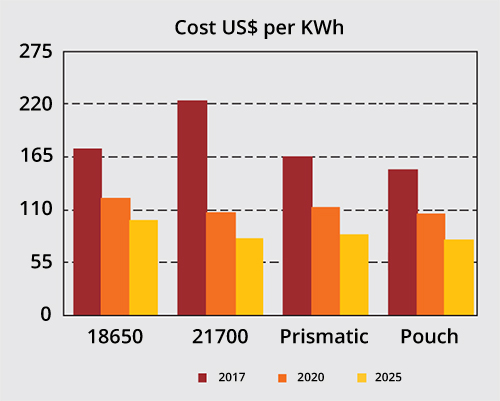
Figure 9: Price comparison of Li-ion cell types.
Automation enables price equilibrium of the 21700 with the pouch cell in 2025. This does not include packaging where the prismatic and pouch cells have a cost advantages.
Source: Battery Experts Forum
Fraunhofer predicts the fastest growth with the 21700 and the pouch cell while the popular 18650 will hold its own. Costs per kWh do not include BMS and packaging. The type cell chosen varies packaging costs as prismatic can easily be stacked; pouch cells may require some compression and cylindrical cells need support systems that create voids. Large packs for electric vehicle also include climate control that adds to cost.
Summary
With the pouch cell, the manufacturer is attempting to simplify cell manufacturing by replicating the packaging of food. Each format has pros and cons as summarized below.
- Cylindrical cell has high specific energy, good mechanical stability and lends itself to automated manufacturing. Cell design allows added safety features that are not possible with other formats (see BU-304b: Making Lithium-ion Safe); it cycles well, offers a long calendar life and is low cost, but it has less than ideal packaging density. The cylindrical cell is commonly used for portable applications.
- Prismatic cell are encased in aluminum or steel for stability. Jelly-rolled or stacked, the cell is space-efficient but can be costlier to manufacture than the cylindrical cell. Modern prismatic cells are used in the electric powertrain and energy storage systems.
- Pouch cell uses laminated architecture in a bag. It is light and cost-effective but exposure to humidity and high temperature can shorten life. Adding a light stack pressure prolongs longevity by preventing delamination. Swelling of 8–10 percent over 500 cycles must be considered with some cell designs. Large cells work best with light loading and moderate charge times. The pouch cell is growing in popularity and serves similar applications to the prismatic cell.
Last Updated 2019-04-24
Make sure to charge the battery every 8 to 10 weeks in order to keep the cells well equalized and balanced, and ensure that the battery is operable. The PCM pulls a small amount of power to equalize the cells and requires periodic charging to ensure continued operation of the battery.
Almost all battery packs, internally, are a combination of several cells connected in series and parallel.
Likewise, several battery packs can be connected in series or parallel for higher voltages and higher capacities.
We do not recommend connecting multiple lithium batteries in series or parallel because Li-ion batteries have internal BMS to protect the battery, and these BMS cannot reliably protect the entire battery pack.
- The lithium batteries we sell are specially designed to be combined in series and parallel, but special attention is needed.
- Lithium batteries with separate charging and discharging ports cannot be used in parallel.
- Batteries should be charged before connecting in parallel so that the voltage of each battery is close.
- The total voltage of the battery pack in series should not exceed the maximum voltage allowed for each battery in series.
- The rated operating current of a battery pack connected in series should be derated to lower than the rated operating current of each battery.
- The higher the voltage of the series connection, the smaller the rated operating current should be, and the large operating current will easily damage the BMS.
- Batteries should be fully charged independently before being connected in series to achieve a balance of charge apexes. Each battery in series shall be fully charged individually every six months to one year to maintain the capacity of the battery pack.
We recommends that you purchase a single battery that meets your voltage and capacity requirement to avoid any potential problems. Problems may arise if you attempt assembly of a larger capacity battery from multiple smaller units. The risks incurred may result in injury depending on output of battery. It is important to understand that LFP batteries are not like Legos – you should not attempt to wire them together as such. Our batteries are all computer controlled and should not be confused with the more common SLA, AA, AAA, and other such basic batteries which do not have any type of computer chip control. If your requirements should fall beyond our standard product lines, we have the capability of manufacturing most custom orders. Our experienced staff will assist you to the best of our abilities.
Make sure the load is not exceeding the rated continuous output current. If the electrical load exceeds the limits of the PCM, the PCM will shut down the pack. To reset, disconnect the electrical load and troubleshoot your load and make sure that the continuous current is less than the maximum continuous current for the pack. To reset the pack, attach the charger back for a few seconds to the battery. If you need a battery with additional current output, please contact us at sales@arbattery.com or call us.
Please contact us at sales@tobattery.com
Don't worry. We encourage you to call because we is uniquely committed to finding the perfect solution for your application because every application has its own unique demands. This extends to consultation of application and any other additional or ancillary inquiries.
- First of all, you must get to know your device:
- What is device's input voltage? ( V)
- What is its power consumption ( Wattage ) ?
- What is Maximum current drain ( A) ?
- What is your expected running time by a battery pack ?
- Decide battery pack voltage you will choose
- Battery pack voltage must be equal or a little higher than your device’s need.
- If you need a exact voltage, which battery pack can not provide, you may consider to use a DC-DC regulator
- Decide Battery pack's Capacity ( mAh or Ah)
- Battery capacity is depended on how long you need to run your device ( hours ), which can be calculated as the follow:
- ( Ah) = Device's Wattage (W) x Time to run ( Hours) / Battery Voltage (V)
- For example, if using a 12V battery pack, and run 100W DC device for 10 hours, you need choose a battery pack with capacity > 83Ah, e.g (100x10)/12 = 83
- 1000 mAh = Ah, higher mAh will provide longer running time.
- Decide Battery pack’s Max. discharging current
- Before order battery pack, you must pay attention on battery pack’s Max. discharging rating on the specification or description. Please don’t think any battery can take any current drain.
- You shall know your device’s max. discharging current. If you don’t know , you must measure it by a multi-meter.
- The battery pack’s Max. discharging rate of the chosen Battery must be higher than that device requires
- Choose Li-Ion, NiMH or Lead acid battery?
- What type of rechargeable battery will work for you. Question is size and weight. The table below lists the advantages and disadvantages of these batteries for your reference
Chemistry | Cost | Weight | Temp(ºC) | Cycle Life | Shelf Life (months) | Volts / Cell |
Lead Acid | $$ | Very Heavy | -65 to 80 | 300 | 12 | 2.0 |
Nickel Cadmium | $$ | Heavy | -20 to 65 | 500+ | 6 | 1.2 |
Nickel Metal Hydride | $$$ | Moderate | -10 to 65 | 500 | 12 | 1.2 |
Lithium Ion | $$$$ | Very Light | -20 to 60 | 500 | 12 | 3.7 |
Lithium Polymer | $$$$$ | Very Light | -20 to 60 | 800 | 12 | 3.7 |
Lithium iron phosphate(LiFePO4) | $$$ | Light | -10 to 60 | 2000 | 12 | 3.2 |
- Li-Ion batteries has the highest energy density ( mAh/weight), and become more and more popular. However, Li-Ion may explode and cause a fire if it dose not use properly.
- Choose a Battery Charger
- Please always follow our suggestion to choose a smart charger. So called smart charger must has automatic cut-off function when battery is full.
- Never use NiMH/Li-ion battery charger for Li-ion/Nimh battery pack, it will cause battery exploded
- Always charging your battery with attention, and put battery in a fire-proof container box to avoid accident.
- Understand risk to use Li-ion battery.

















.jpg)
.jpg)
.jpg)
.jpg)
.jpg)

.jpg)